 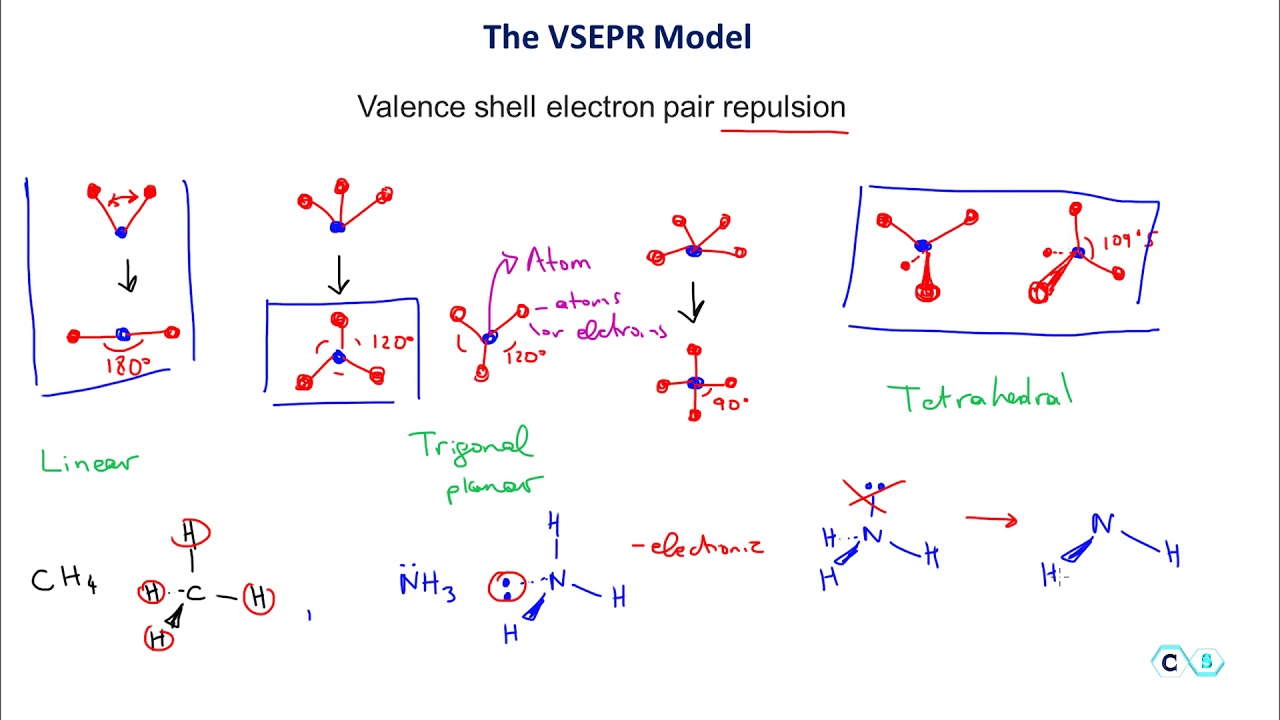
The VSEPR model has been an educational tool more than a research tool, but its value as the latter should not be ignored. If this is true to any extent, and I believe it is to a great extent, it has been beneficial to mankind. Yet Gillespie’s model certainly made its important aspects more user-friendly. 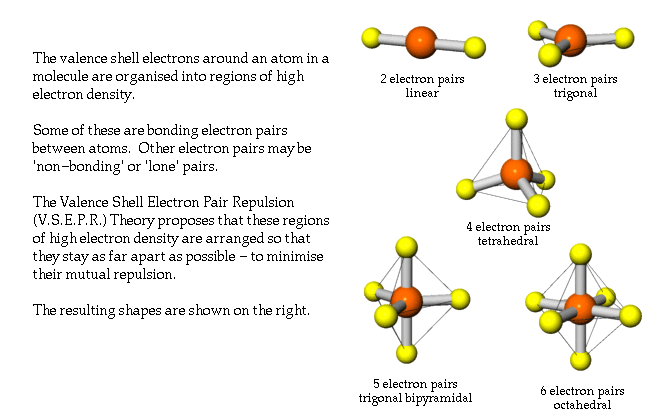
Chemistry is not an easy subject neither is it among the favorite ones for most pupils. For chemistry it sounds, “to the person who shall have made the most important chemical discovery or improvement.” The VSEPR model has benefited mankind in facilitating the dissemination of chemical literacy, and it was a discovery or at least an improvement of significance. The other is specific to each of the five categories although the distinctions are slight. One is a general stipulation referring to having conferred the greatest benefit on mankind. The criteria for the Prize according to Alfred Nobel’s Will have double requirements. In fact, it could have been among the recently rather rare pure chemistry awards countering the growing tendency of using chemistry Nobel awards for achievements in discoveries of biological importance. Although the Nobel Prize should not be made into any kind of absolute measure, in a way it has become one, and had Gillespie’s VSEPR model been awarded one, I doubt it would have elicited any protest. It has certainly made it easier for students at any level of chemistry instruction to grasp the notion of and variations in molecular structure, meaning the geometry of molecules in this case. It is very difficult if at all possible to assess the importance of the VSEPR model in education. One is in chemistry education and the other is as a research tool. The VSEPR model has had double significance. I was taken by this model so much that I penned a long letter to my wife-and-graduate student back in Budapest, and illustrated it with loops of electron pairs in color (the new knowledge of this attractive model kept me up all night following my seminar, or it may have been just jet leg). I was trying to observe trends in the structural variations and even interpret them, when Russ asked whether I had tried to apply VSEPR considerations to them. I talked about the structures of some simple molecules containing sulfur as we determined them by gas-phase electron diffraction in Budapest. It was in January 1969 I was on my way to the University of Texas at Austin, and was invited to Indiana University in Bloomington by Russel A. I was introduced to the model in the discussion following my first ever seminar presentation in the United States. Once in possession of the model, many of the structural variations could be inscribed into the realm of a set of rudimentary rules. This model then was augmented with a few principal rules pertaining to the effects of multiple bonds, ligand electronegativity, and d-orbital availability, but its basic tenet has remained its cornerstone.Īs a student, I had no knowledge of this model, and much of the information about the shapes of molecules we had to learn one by one. The essence of the model is that the repulsion interactions of all electron pairs, counting both shared and unshared ones, in the valence shell of the central atom in a molecule determines the shape of the molecule. The discovery of the VSEPR model was not inevitable and, although it relied on previous notions of trends in the variations in inorganic molecular structures, it was more unique and more personal than most scientific discoveries. Ron’s great contribution with the Valence Shell Electron Pair Repulsion or VSEPR model (initially together with Ronald Nyholm, who soon died, much prematurely) is of a different character. Also, if one scientist does not make a certain discovery, another will, sooner or later. However, research achievements are almost always parts of a process they build on previous achievements, on the one hand, and are superseded by others, on the other hand. 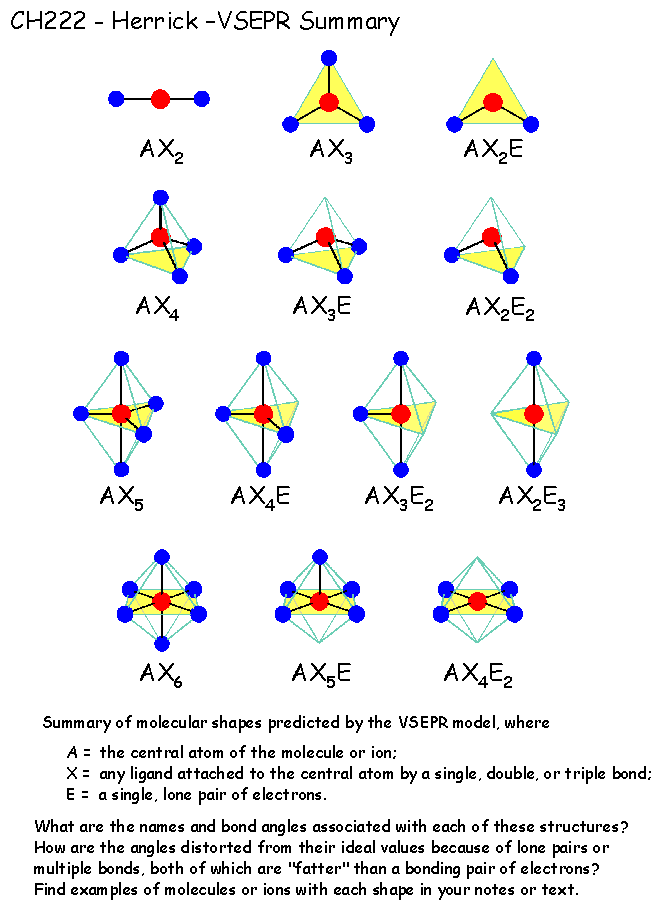
Although it did not, nobody doubts that his contribution was of Nobel-prize-caliber. His pioneering work in superacids has been recognized and might have even earned him a share of the Nobel Prize. Ron-as he is known to many-has been both a great research chemist and a chemistry educator. The community of chemists-in particular in inorganic chemistry and structural chemistry-honors him in many ways. 1) of superacid and VSEPR fame is 85 years old this year. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
